- Blog
- Stellar phoenix photo recovery 7 key download
- Mini excavator buckets
- Flash exporter for clickteam fusion 2-5 download
- Vision credit union car loan payment calculator
- Hap codec for after effects download
- Time zone map time zone map
- Dektop cpu and gpu temp monitor
- Best capture one presets download free
- TeamViewer APKqwondershare apk pro torrent download
- Boot camp windows mac os
- Automatic pool covers westfield indiana
- Soulcalibur iv usa ps3 dlc pkg
- Anylogic tutorial slider
- Logo maker for youtube
- Picasa 3 free download for windows 10
- Keystrokes mod forge 1-8-9
- Chal jivi laiye full movie download 2019
- House of highlights creator league schedule
- Creamy vanilla fudge recipe
- Invisible item frame minecraft command
- Fix lath and plaster wall
- Php javascript excel type grids
- Youtube downloader pc windows 7
- Color coded periodic table alkali metals
- Google cloud ftp into server
- Daymap salisbury high school
- Keyshot 7 education download crack
- Free marker script font
- Drexel studio furniture catalog
- Best ipad budget planner
- Calculate statistics linear regression equation
- Private aural training lesoons advertisement picture
- Bible studies elijah
- Printable check register to fit checkbook
- Free key codes for xplane 11
- Vmware player enhanced keyboard driver
- Doppler radar mexico and southwestern united states
- Billy get grounded
- Hollywood movie hindi audio track blogspot
- Google app closest to windows classic solitaire
- Download driver notebook acer aspire v5 131
- Southeast united states doppler radar
- Games online 2048
- Installing omnisphere 2 cracked
- Affinity photo plugins 2018
- Play geometry dash free the impossible game
:max_bytes(150000):strip_icc()/PeriodicTableCrystal-56a12d9b5f9b58b7d0bccfdf.png)
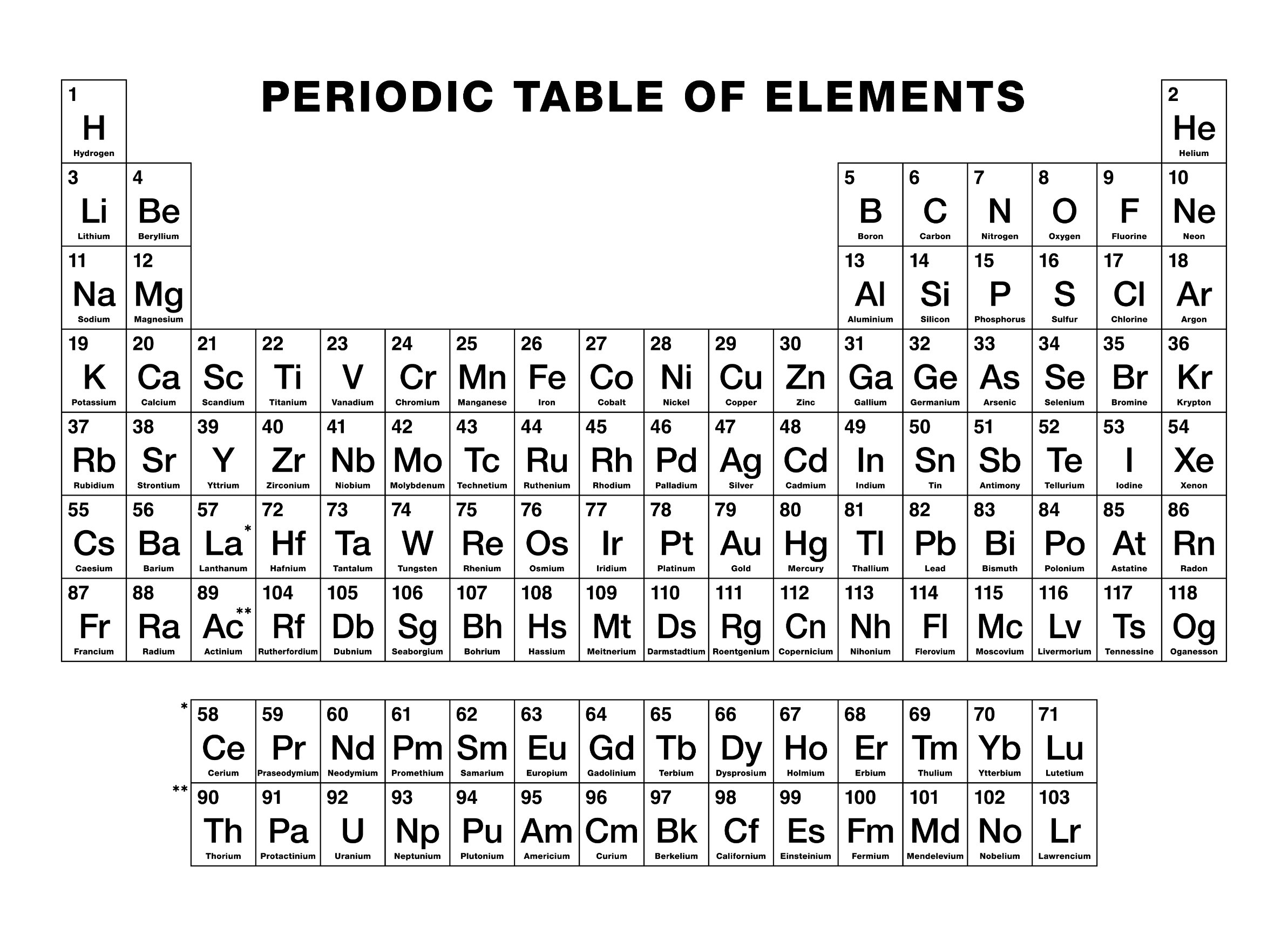
Outline the symbol’s box in dark green if it is RADIOACTIVE in its most common form.Use the following chart to color the periodic table.Place the atomic number for each element above the symbol.Write the symbol of each element that is a man-made element as an outline.Write the symbol of each element that is a liquid at ordinary condition in BLUE.Write the symbol of each element that is a solid at ordinary conditions in BLACK.Write the symbol of each element that exists as a gas at ordinary conditions in RED.Write the names of the two rows at the bottom of the chart: lanthanides and actinides.Group 3-12 (collectively) transition metals Write the name of each of the following groups above the number:.Draw a heavy lack line between the metals and nonmetals.So, I'm wondering what other people's opinions on the matter is.Objective: To be able to use the periodic table to identify and classify elements and to use the periodic table to predict the behavior of elements For instance, when alkali metals burn in air, the observed products are Li 2 O (white), Na 2 O 2 (pale yellow), KO 2 (orange), RbO 2 (brown), and. I think hydrogen should just be in its own group (since when its electron shell is filled, there are only 2 electrons instead of 8), with it being the most similar to alkali metals. Reacting the alkali metals with oxygen, the lightest element in group 16, is more complex, and the stoichiometry of the product depends on both the metal:oxygen ratio and the size of the metal atom. It is also much less electropositive than lithium but only about as electronegative as iodine (in fact, I think iodine is more electronegative). The second group of s-block has the alkali earth metals. It has the same most common charge (+1) as all alkali metals, is mainly reducing like them, and reacts with halogens and oxygen vigorously, along with a few other examples.īut a few of its properties doesn't fit with either the halogens or the alkali metals. Alkaline earth metals are also present at the top left of the periodic table. For example, it forms stable/meta-stable peroxides/superoxides, like alkali metals, particularly potassium and beyond. Hydrogen fits in its current position on the periodic table (group 1) much better than in group 17 (not a perfect fit, though). The period table template provides information about the elements, their groups, and the various trends they show. It is also reactive with non-metals like oxygen, but not with metals like iron (still reacts slowly though, to form hydrides), unlike all halogens. An oxidation state of +1 is very unstable in all halogens. Second, it doesn't even behave like a halogen, being mainly in the +1 oxidation state (doesn't happen to halogens, the most stable compounds of halogens have either -1 or a very high oxidation state, like +7 or +5.
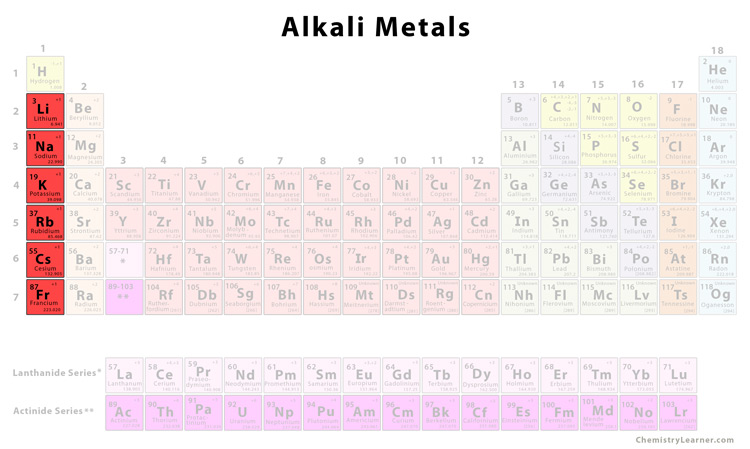
Hydrogen would have to be more electronegative than fluorine should it be a halogen. Firstly, electronegativity decreases as you go down the periodic table, and the element becomes more and more metallic. Whilst hydrogen do not exactly fit in group 1 as an "alkali metal" (it's a gas instead of a metal, except in special conditions, it forms negatively-charged ions, and it is far less electropositive than lithium), it absolutely cannot be a halogen. Are you are looking for lots of last minute lessons on the periodic graphic, you’ve found an article that will help you. High school and median college students will appreciate of tactic background the color coding. This site ( ) claims that hydrogen should actually be put in group 17 instead of group 1, basically claiming that hydrogen is a halogen. There are 7 lessons you capacity teach with color pencils and a blank periodic table.
